Description
Millions of people worldwide suffer from osteoporosis, which is characterised by weakening bones that are prone to fractures. It’s a quiet yet devastating condition that goes undetected until a fracture occurs. As bone density declines, the risk of fractures increases, affecting everyday living and general well-being. Individuals may battle osteoporosis and maintain bone health by taking preventative steps and using effective therapies such as Osteofos 35 mg.
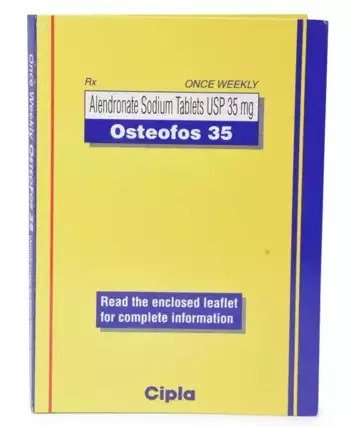
What is Osteofos 35 mg?
Osteofos 35 Mg is a powerful drug that is particularly intended to improve bone density and minimise fracture risk in osteoporosis patients. It is a bisphosphonate medication, which works by suppressing bone breakdown and thereby conserving bone mass and strength. The active component, alendronate sodium, promotes bone health by delaying bone resorption.
How Does Osteofos 35 Mg work?
Alendronate sodium in Osteofos 35 Mg acts by binding to bone tissue, especially in regions of active resorption. It contributes to bone density and structure preservation by reducing osteoclast activity, which is responsible for bone breakdown. This method efficiently lowers the risk of fractures while improving overall bone health.
Benefits of Osteofos 35mg
1. Increased Bone Density: Studies have shown that regular usage of Osteofos 35 Mg improves bone mineral density, strengthens bones, and lowers fracture susceptibility.
2. Fracture Prevention: By slowing bone resorption, Osteofos 35 Mg greatly reduces the incidence of fractures, particularly in high-risk people.
3. Long-term Bone Health: Continued use of Osteofos 35 Mg provides long-term advantages by conserving bone mass and avoiding further degeneration.






Reviews
There are no reviews yet.